 The periodic table as a list of elements arranged so as to demonstrate trends in their physical and chemical properties.Describe and model the structure of the atom in terms of the nucleus, protons, neutrons and electrons comparing mass and charge of protons neutrond and electrons. Use the Periodic Table to predict the ratio of atoms in compounds of two elements. Unit 1: Structures, Trends, Chemical Reactions, Quantitative Chemistry and Analysis.Unit C1: Structures, Trends, Chemical Reactions, Quantitative Chemistry and Analysis.(g) elements being arranged in order of increasing atomic number and in groups and periods in the modern Periodic Table, with elements having similar properties appearing in the same groups.2.2 ATOMIC STRUCTURE AND THE PERIODIC TABLE.(h) elements being arranged in order of increasing atomic number and in groups and periods in the modern Periodic Table, with elements having similar properties appearing in the same groups.1.2 ATOMIC STRUCTURE AND THE PERIODIC TABLE.Unit 1: CHEMICAL SUBSTANCES, REACTIONS and ESSENTIAL RESOURCES.(a) elements being arranged according to atomic number in the Periodic Table.  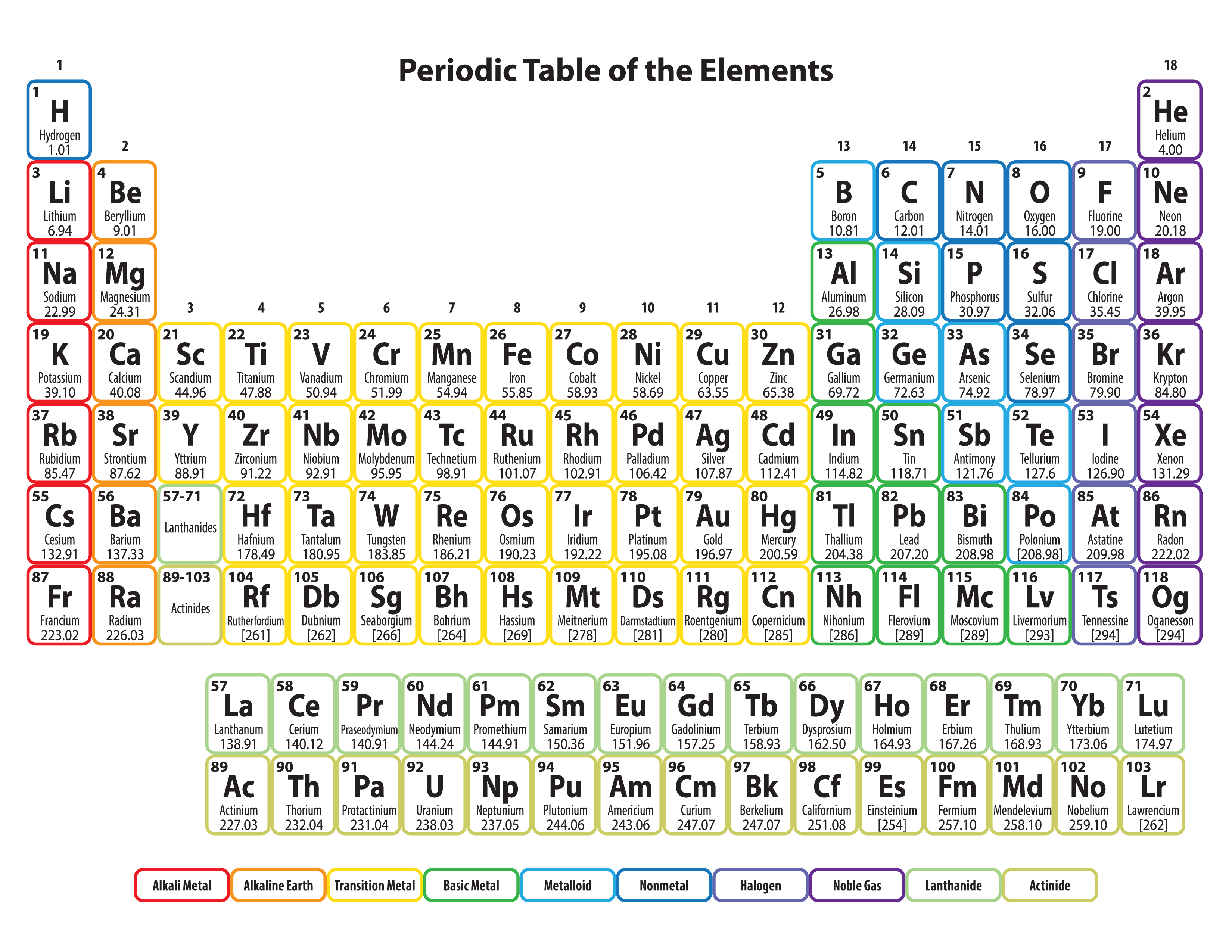
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
/accurate-illustration-of-the-periodic-table-82020791-57cc76f23df78c71b66efbd7.jpg)
 RSS Feed
RSS Feed
